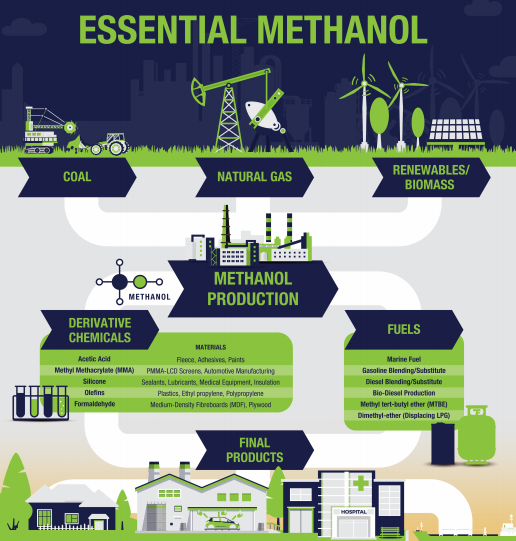
Methanol is a clear liquid chemical used in thousands of everyday products, including plastics, paints, cosmetics, and fuels. Methanol is also an energy resource used in the marine, automotive, and electricity sectors and an emerging renewable energy resource.
Methanol (CH3OH) is water-soluble and readily biodegradable, comprising four parts hydrogen, one part oxygen, and one part carbon, and is the simplest member of a group of organic chemicals called alcohols. Methanol is a clean-burning, biodegradable fuel. Increasingly, methanol’s environmental and economic advantages make it an attractive alternative fuel for powering vehicles and ships, cooking food, and heating homes.
Typically, methanol is produced with natural gas. However, with increasing interest in sustainability, methanol has proven versatile in its production pathways. More projects are utilizing renewable feedstocks such as agricultural waste, Municipal Solid Waste (MSW), sewage, renewable electricity, and captured CO2.
Methanol production with renewable feedstocks significantly reduces greenhouse gas intensity and contributes to the energy transition in end-user markets. As a building block for many downstream chemicals, sustainably produced methanol facilitates the growth of the green chemicals sector. Sustainably produced methanol is also being utilized as an alternative drop-in fuel in shipping, road transport, and power generation to lower overall emissions and impact on the environment.

Physical Properties
Methanol Technical Data Sheet
The Methanol Institute produced a Methanol Technical Data Sheet describing methanol’s physical properties and key applications.
IMPCA Methanol Reference Specifications
The International Methanol Producers & Consumers Association’s (IMPCA) Global Methanol Reference Specification.
Physical properties of Pure Methanol
A chart detailing the physical properties of pure (neat) methanol.
Boiling and Flash Points of Methanol
A chart that demonstrates the boiling and flash points for methanol and methanol-water solutions.
Freezing Points of Methanol
A chart that shows the relative freezing points for methanol and methanol-water solutions.
Water-Methanol Mix Densities
A chart that shows the relative density of water-methanol mixes
Latent Heat and Vaporization
A chart that shows the latent heat of vaporization and specific heat of liquid methanol
Material Compatibility
Compatibility of Elastomers in Neat Methanol Service
A look into elastomer and rubber materials which help prevent leakage of methanol liquid and vapors into the surrounding environment.
Compatibility of Metal Alloys in Neat Methanol Service
Detailed technical bulletin describing the appropriate metals and alloys for various applications
Materials Selection for Neat Methanol Service
An overall summary of material selection including information on standards, toxicity, and safety.